 
3) Change in Number of Particles (especially for Gases) 4) Mixing of Particles. What factors affect entropy 1) Change in Phase (Physical State) 2) Change in Temperature. $^*$ For the concept to discern calorimetric entropy from spectroscopic entropy, see for example this Chemistry LibreText an literature example e.g., in 1958JAmChemSoc1054. Which substance has lowest entropy Answer and Explanation: Solids have the lowest entropy value among the other states of matter since the molecules in solids are stationary. Thermal equilibrium will be reached at the maximum entropy state (Smax)TE. An increase in the number of moles on the product side means higher entropy. If the water is replaced with warm nitrogen at 350 K, heat transfer will. The other two gases have the same number of. A decrease in the number of moles on the product side means lower entropy. As a result, because NO 2 contains the most atoms, it has the most vibration modes and the highest molar entropy. I feel like the answer is something simple that I have missed. How do you know if a reaction has high entropy Therefore, if the reaction involves only gases, the entropy is related to the total number of moles on either side of the reaction. Figure 16.10 The entropy of a substance increases (S > 0) as it transforms from a relatively ordered solid, to a less-ordered liquid, and then to a still less-ordered gas. 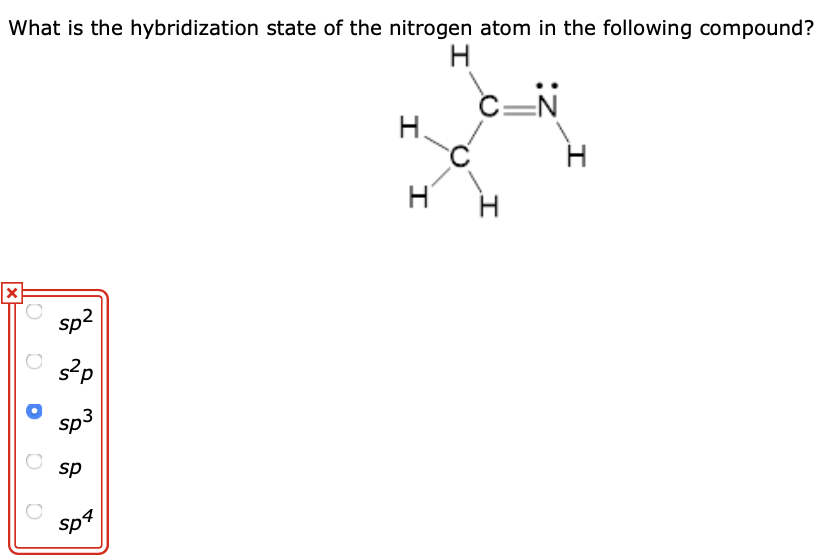
So where does the difference in the calculated entropy come from? (I think the excited states are all inaccessible at room temperature). The electronic term should also be zero in both cases, as the electronic partition function would be 1 for both. This is because gases can exist in a large number of different microstates. 
The rotational and vibrational terms are assumed to be same in the question. While solving a question set, I found a question with this data table (at $\pu$. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
